MIMIC-KeY
in a nutshell
OBJECTIVE 1:
KEY UNDERSTANDING
WHAT?
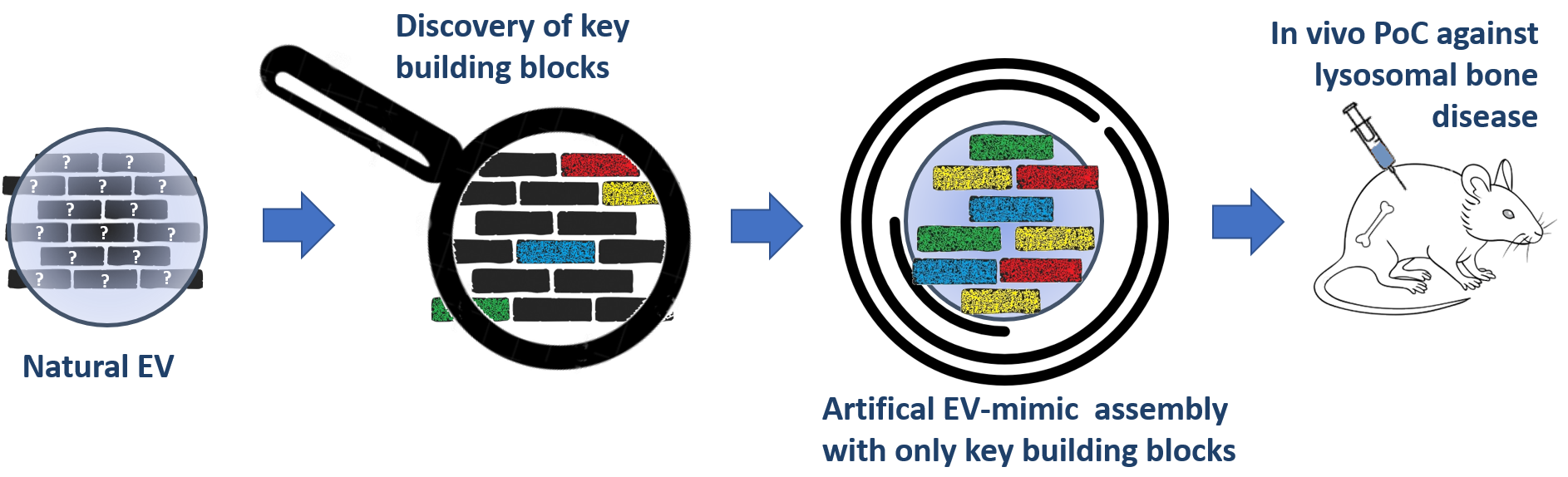
We aim to learn from nature and identify the key molecules at the surface of natural EVs (derived from cancer cells), that enable their natural homing capability and their content transfer to the target recipient cells.
WHY?
EVs should display membrane proteins that target bone or enable the transfer of the oncogenic content to the bone cells.
Focus into cancer tissues that provoke metastasis in the bone to learn how to treat an unmet clinical need
HOW?
We start from prostate cancer and multiple myeloma, since their related EVs show a strong tendency to prepare a metastatic niche in the bone (osteoclasts cells). We aim to treat lysosomal bone metabolic disease (defective osteoclasts)
Thanks to a synergistic combination of ground-breaking and multidisciplinary approaches: multiscale simulations, biological proteomic profiling, novel methods in flow cytometry and super-resolution imaging.
OBJECTIVE 2:
MIMIC
WHAT?
We aim to synthetically produce new artificial EV-mimics, made by synthetic and supramolecular nanoparticles, NPs, and loaded with the desired therapeutic cargo (enzyme Cathepsin K, CTSK).
WHY?
Artificial EV-mimics will behave like natural metastatic ones, with the advantage of being fully artificial with uniform size and displaying only the essential key molecules
HOW?
We will create artificial EVs with the minimum requirement for cell recognition and cargo transfer, composed of: artificial lipid bilayers/functional proteins, triggerable porous organosilica nanocapsules and soft nanogels, with perfect control of size and monodispersity.
They will be able to control parameters influencing adhesion and mobility of lipid bilayers to metal oxide NP, transport relevant therapeutics, control cargo release by engineering the destruction of the carrier, target metabolic bone disease.
Work Package organization
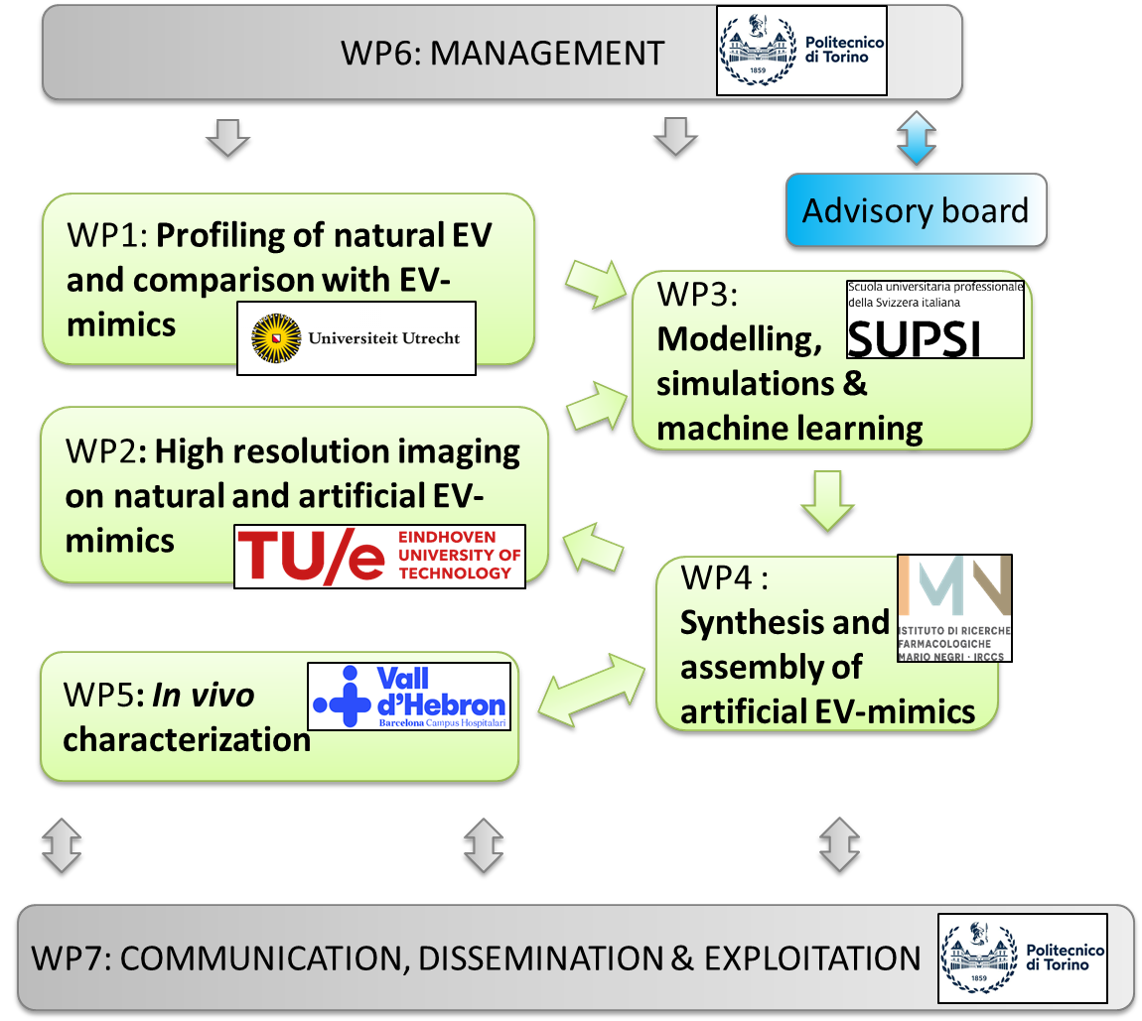
WP1
Profiling of bone-targeting natural EVs and functional comparison of natural EVs and EV-mimics
Objectives :
1) Identify key molecular components enabling natural EVs to bind and transfer therapeutic cargo to specific bone cells;
2) Assess whether EV-mimics designed for bone targeting resemble natural EVs in terms of cell binding and cargo delivery capacity.
WP2

High resolution imaging of natural and artificial EV-mimics
Objectives:
1) Enable emerging imaging technologies for EVs characterization;
2) Obtain a map of protein and lipids in EVs at the single-molecule and single-vesicle level;
3) Characterize the artificial EVs and compare them with the natural ones.
WP3
Modelling, simulations & machine learning
Objectives:
1) Develop multiscale models and use molecular simulations for studying lipid self-assembly on the surface of hard-core NPs;
2) Submolecular-resolution characterization of structure and dynamics of the EVs (lipid envelope);
3) Develop statistical models to predict the dynamic enhancement of multivalent molecular recognition between the EVs and EV-mimics to target cell surface models.
WP4
Synthesis and assembly of artificial EV-mimics
Objectives:
1) Create artificial EV mimicking the natural ones equipped with the receptors identified in WP1 and WP2 as targeting units;
2) Compare the natural EVs and the artificial ones in the targeting behaviour;
3) Obtain a quality control to know that the essential EV proteins/lipids are on the same particle as the therapeutic cargo.
WP5

In vivo characterization
Objectives:
1) In vivo toxicity and biodistribution studies of natural EV containing CTSK and RANKL in healthy mice;
2) In vivo toxicity and biodistribution studies in ctsk-null mice model; 3) Proof of concept approach of artificial EV-mimics in in vivo relevant application, i.e. lysosomal metabolic bone disease
WP6&7

Management, Communication, Dissemination, Exploitation
Objectives:
1) Ensure the overall coordination and day-to-day management of the project, including the overall project plan;
2) Handle all the financial, legal and contractual matters.
3) Ensure ongoing communication, dissemination and exploitation of the project’s results.
4) Handle all matters in intellectual property and data management.

